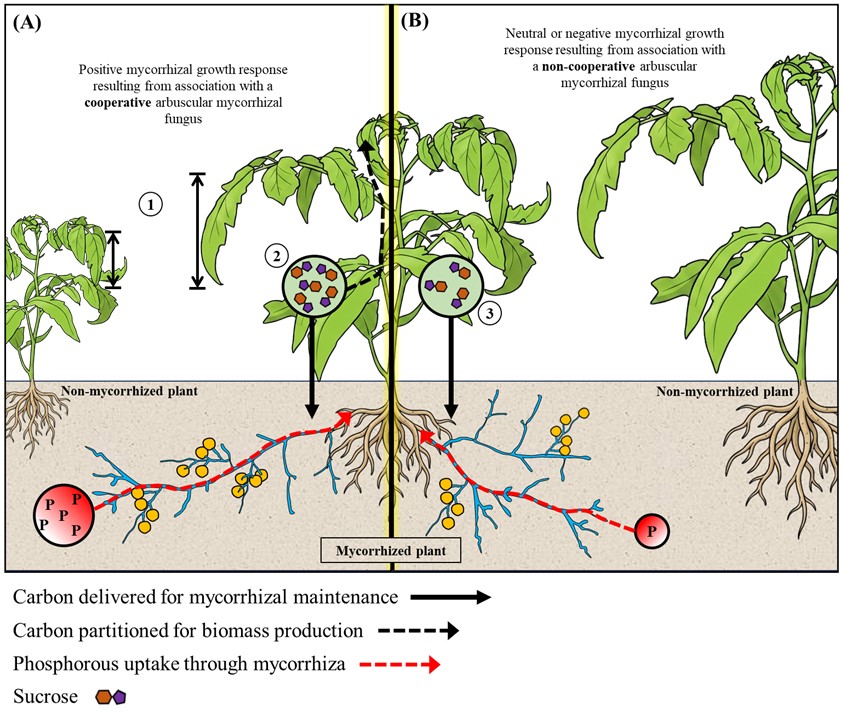
Dynamics of plants source – sink tissue relationships modulated by arbuscular mycorrhizal symbiosis
Abstract
Keywords
Full Text:
PDFReferences
An, J., Zeng, T., Ji, C., de Graaf, S., Zheng, Z., Xiao, T.T., Deng, X., Xiao, S., Bisseling, T., Limpens, E. and Pan, Z., 2019. A Medicago truncatula SWEET transporter implicated in arbuscule maintenance during arbuscular mycorrhizal symbiosis. New Phytologist, 224(1), pp.396–408. https://doi.org/10.1111/nph.15975
Andrino, A., Guggenberger, G., Sauheitl, L., Burkart, S. and Boy, J., 2021. Carbon investment into mobilization of mineral and organic phosphorus by arbuscular mycorrhiza. Biology and Fertility of Soils, 57(1), pp.47–64. https://doi.org/10.1007/s00374-020-01505-5
Bago, B. and Bécard, G., 2002. Bases of the obligate biotrophy of arbuscular mycorrhizal fungi. In: Mycorrhizal Technology in Agriculture. [online] Basel: Birkhäuser Basel. pp.33–48. https://doi.org/10.1007/978-3-0348-8117-3_3
Bago, B., Cano, C., Azcón-Aguilar, C., Samson, J., Coughlan, A.P. and Piché, Y., 2004. Differential morphogenesis of the extraradical mycelium of an arbuscular mycorrhizal fungus grown monoxenically on spatially heterogeneous culture media. Mycologia, 96(3), pp.452–462. https://doi.org/10.1080/15572536.2005.11832944
Bago, B., Pfeffer, P.E., Abubaker, J., Jun, J., Allen, J.W., Brouillette, J., Douds, D.D., Lammers, P.J. and Shachar-Hill, Y., 2003. Carbon export from arbuscular mycorrhizal roots involves the translocation of carbohydrate as well as lipid. Plant Physiology, 131(3), pp.1496–1507. https://doi.org/10.1104/pp.102.007765
Bago, B., Pfeffer, P.E., Douds, D.D., Brouillette, J., Bécard, G. and Shachar-Hill, Y., 1999. Carbon metabolism in spores of the arbuscular mycorrhizal fungus Glomus intraradices as revealed by nuclear magnetic resonance spectroscopy. Plant Physiology, 121(1), pp.263–271. https://doi.org/10.1104/pp.121.1.263
Baier, M.C., Keck, M., Gödde, V., Niehaus, K., Küster, H. and Hohnjec, N., 2010. Knockdown of the symbiotic sucrose synthase MtSucS1 affects arbuscule maturation and maintenance in mycorrhizal roots of Medicago truncatula . Plant Physiology, 152(2), pp.1000–1014. https://doi.org/10.1104/pp.109.149898
Balzergue, C., Chabaud, M., Barker, D. G., Bécard, G., and Rochange, S. F. 2013. High phosphate reduces host ability to develop arbuscular mycorrhizal symbiosis without affecting root calcium spiking responses to the fungus. Frontiers in Plant Science, 4, 426. https://doi.org/10.3389/fpls.2013.00426
Besserer, A., Puech-Pagès, V., Kiefer, P., Gomez-Roldan, V., Jauneau, A., Roy, S., Portais, J.C., Roux, C., Bécard, G. and Séjalon-Delmas, N., 2006. Strigolactones stimulate arbuscular mycorrhizal fungi by activating mitochondria. PLoS Biology, 4(7), pp.1239–1247. https://doi.org/10.1371/journal.pbio.0040226
Bihmidine, S., Hunter, C.T., Johns, C.E., Koch, K.E. and Braun, D.M., 2013. Regulation of assimilate import into sink organs: Update on molecular drivers of sink strength. Frontiers in Plant Science, 4, pp.1–15. https://doi.org/10.3389/fpls.2013.00177
Bilska-Kos, A., Mytych, J., Suski, S., Mago?, J., Ochodzki, P. and Zebrowski, J., 2020. Sucrose phosphate synthase (SPS), sucrose synthase (SUS) and their products in the leaves of Miscanthus × giganteus and Zea mays at low temperature. Planta, 252(2), pp.1–14. https://doi.org/10.1007/s00425-020-03421-2
Bitterlich, M., Krügel, U., Boldt-Burisch, K., Franken, P. and Kühn, C., 2014. The sucrose transporter SlSUT2 from tomato interacts with brassinosteroid functioning and affects arbuscular mycorrhiza formation. Plant Journal, 78(5), pp.877–889. https://doi.org/10.1111/tpj.12515
Boldt, K., Pörs, Y., Haupt, B., Bitterlich, M., Kühn, C., Grimm, B. and Franken, P., 2011. Photochemical processes, carbon assimilation and RNA accumulation of sucrose transporter genes in tomato arbuscular mycorrhiza. Journal of Plant Physiology, 168(11), pp.1256–1263. https://doi.org/10.1016/j.jplph.2011.01.026
Bonfante, P., 2018. The future has roots in the past: the ideas and scientists that shaped mycorrhizal research. New Phytologist, 220(4), pp.982–995. https://doi.org/10.1111/nph.15397
Bunn, R.A., Corrêa, A., Joshi, J., Kaiser, C., Lekberg, Y., Prescott, C.E., Sala, A. and Karst, J., 2024. What determines transfer of carbon from plants to mycorrhizal fungi? New Phytologist, 244(4), pp.1199–1215. https://doi.org/10.1111/nph.20145
Cargill, R.I.M., Shimizu, T.S., Kiers, E.T. and Kokkoris, V., 2025. Cellular anatomy of arbuscular mycorrhizal fungi. Current Biology, 35(11), pp.R545–R562. https://doi.org/10.1016/j.cub.2025.03.053
Cesaro, P., Massa, N., Cantamessa, S., Todeschini, V., Bona, E., Berta, G., Barbato, R. and Lingua, G., 2020. Tomato responses to Funneliformis mosseae during the early stages of arbuscular mycorrhizal symbiosis. Mycorrhiza, 30(5), pp.601–610. https://doi.org/10.1007/s00572-020-00973-9
Chávez-Bárcenas, A.T., Valdez-Alarcón, J.J., Mart??nez-Trujillo, M., Chen, L., Xoconostle-Cázares, B., Lucas, W.J. and Herrera-Estrella, L., 2000. Tissue-specific and developmental pattern of expression of the rice sps1 gene. Plant Physiology, 124(2), pp.641–654. https://doi.org/10.1104/pp.124.2.641
Chen, L.-Q., Qu, X.-Q., Hou, B.H., Sosso, D., Osorio, S., Fernie, A.R. and Frommer, W.B., 2012. Sucrose efflux mediated by SWEET proteins as a key step for pholem transport. Science, 335(6065), pp.207–211. https://doi.org/10.1007/s10886-010-9780-8
Chen, L.Q., 2014. SWEET sugar transporters for phloem transport and pathogen nutrition. New Phytologist, 201(4), pp.1150–1155. https://doi.org/10.1111/nph.12445
Chen, S., Hajirezaei, M. and Börnke, F., 2005. Differential expression of sucrose-phosphate synthase isoenzymes in tobacco reflects their functional specialization during dark-governed starch mobilization in source leaves. Plant Physiology, 139(3), pp.1163–1174. https://doi.org/10.1104/pp.105.069468
Chen, Z., Gao, K., Su, X., Rao, P. and An, X., 2015. Genome-wide identification of the invertase gene family in Populus. PLoS ONE, 10(9), pp.1–21. https://doi.org/10.1371/journal.pone.0138540
Das, D., Paries, M., Hobecker, K., Gigl, M., Dawid, C., Lam, H.M., Zhang, J., Chen, M. and Gutjahr, C., 2022. PHOSPHATE STARVATION RESPONSE transcription factors enable arbuscular mycorrhiza symbiosis. Nature Communications, 13(1). https://doi.org/10.1038/s41467-022-27976-8
Deng, Q., Wang, H., Qiu, Y., Wang, D., Xia, Y., Zhang, Y., Pei, M., Zhao, Y., Xu, X. and Zhang, H., 2025. The multifaceted impact of karrikin signaling in plants. International Journal of Molecular Sciences 26, pp.2775. https://doi.org/10.3390/ijms26062775
Doidy, J., Van Tuinen, D., Lamotte, O., Corneillat, M., Alcaraz, G. and Wipf, D., 2012. The Medicago truncatula sucrose transporter family: Characterization and implication of key members in carbon partitioning towards arbuscular mycorrhizal fungi. Molecular Plant, 5(6), pp.1346–1358. https://doi.org/10.1093/mp/sss079
Dong, S. and Beckles, D.M., 2019. Dynamic changes in the starch-sugar interconversion within plant source and sink tissues promote a better abiotic stress response. Journal of Plant Physiology, 234–235, pp.80–93. https://doi.org/10.1016/j.jplph.2019.01.007
Farrar, J.F., 1993. Sink strength: What is it and how do we measure it? A summary. Plant, Cell & Environment, 16(9), pp.1045–1046. https://doi.org/10.1111/j.1365-3040.1996.tb02061.x
Feng, C.Y., Han, J.X., Han, X.X. and Jiang, J., 2015. Genome-wide identification, phylogeny, and expression analysis of the SWEET gene family in tomato. Gene, 573(2), pp.261–272. https://doi.org/10.1016/j.gene.2015.07.055
Figueiredo, A.F., Boy, J. and Guggenberger, G., 2021. Common mycorrhizae network: A review of the theories and mechanisms behind underground interactions. Frontiers in Fungal Biology, 2, pp.1–13. https://doi.org/10.3389/ffunb.2021.735299
Fiorilli, V., Martínez-Medina, A., Pozo, M.J. and Lanfranco, L., 2024. Plant immunity modulation in arbuscular mycorrhizal symbiosis and its impact on pathogens and pests. Annual Review of Phytopathology, 62(1), pp.127–156. https://doi.org/10.1146/annurev-phyto-121423-042014
Flügge, U.I., Fischer, K., Gross, A., Sebald, W., Lottspeich, F. and Eckerskorn, C., 1989. The triose phosphate-3-phosphoglycerate-phosphate translocator from spinach chloroplasts: nucleotide sequence of a full-length cDNA clone and import of the in vitro synthesized precursor protein into chloroplasts. The EMBO Journal, 8(1), pp.39–46. https://doi.org/10.1002/j.1460-2075.1989.tb03346.x
Foyer, C.H. and Paul, M.J., 2001. Source–Sink Relationships. In: Encyclopedia of Life Sciences. Wiley. pp.365–388. https://doi.org/10.1038/npg.els.0001304
García-Rodríguez, S., Azcón-Aguilar, C. and Ferrol, N., 2007. Transcriptional regulation of host enzymes involved in the cleavage of sucrose during arbuscular mycorrhizal symbiosis. Physiologia Plantarum, 129(4), pp.737–746. https://doi.org/10.1111/j.1399-3054.2007.00873.x
Gavito, M.E., Jakobsen, I., Mikkelsen, T.N. and Mora, F., 2019. Direct evidence for modulation of photosynthesis by an arbuscular mycorrhiza-induced carbon sink strength. New Phytologist, 223(2), pp.896–907. https://doi.org/10.1111/nph.15806
Genre, A., Lanfranco, L., Perotto, S. and Bonfante, P., 2020. Unique and common traits in mycorrhizal symbioses. Nature Reviews Microbiology, 18(11), pp.649–660. https://doi.org/10.1038/s41579-020-0402-3
Glanz-Idan, N. and Wolf, S., 2020. Upregulation of photosynthesis in mineral nutrition-deficient tomato plants by reduced source-to-sink ratio. Plant Signaling and Behavior, 15(2), pp.1712543. https://doi.org/10.1080/15592324.2020.1712543
Graham, J.H. and Abbott, L.K., 2000. Wheat responses to aggressive and non-aggressive arbuscular mycorrhizal fungi. Plant and Soil, 220(1–2), pp.207–218. https://doi.org/10.1023/a:1004709209009
Gutjahr, C., Gobbato, E., Choi, J., Riemann, M., Johnston, M.G., Summers, W., Carbonnel, S., Mansfield, C., Yang, S.Y., Nadal, M., Acosta, I., Takano, M., Jiao, W.B., Schneeberger, K., Kelly, K.A. and Paszkowski, U., 2015. Rice perception of symbiotic arbuscular mycorrhizal fungi requires the karrikin receptor complex. Science, 350(6267), pp.1521–1524. https://doi.org/10.1126/science.aac9715
Gutjahr, C., Novero, M., Welham, T., Wang, T. and Bonfante, P., 2011. Root starch accumulation in response to arbuscular mycorrhizal colonization differs among Lotus japonicus starch mutants. Planta, 234(3), pp.639–646. https://doi.org/10.1007/s00425-011-1472-2
Gutjahr, C., Radovanovic, D., Geoffroy, J., Zhang, Q., Siegler, H., Chiapello, M., Casieri, L., An, K., An, G., Guiderdoni, E., Kumar, C.S., Sundaresan, V., Harrison, M.J. and Paszkowski, U., 2012. The half-size ABC transporters STR1 and STR2 are indispensable for mycorrhizal arbuscule formation in rice. Plant Journal, 69(5), pp.906–920. https://doi.org/10.1111/j.1365-313X.2011.04842.x
Harrison, M.J., 1996. A sugar transporter from Medicago truncatula: altered expression pattern in roots during vesicular?arbuscular (VA) mycorrhizal associations. The Plant Journal, 9(4), pp.491–503. https://doi.org/10.1046/j.1365-313X.1996.09040491.x
Helber, N., Wippel, K., Sauer, N., Schaarschmidt, S., Hause, B. and Requena, N., 2011. A versatile monosaccharide transporter that operates in the arbuscular mycorrhizal fungus Glomus sp is crucial for the symbiotic relationship with plants . The Plant Cell, 23(10), pp.3812–3823. https://doi.org/10.1105/tpc.111.089813
Hennion, N., Durand, M., Vriet, C., Doidy, J., Maurousset, L., Lemoine, R. and Pourtau, N., 2019. Sugars en route to the roots. Transport, metabolism and storage within plant roots and towards microorganisms of the rhizosphere. Physiologia Plantarum, 165(1), pp.44–57. https://doi.org/10.1111/ppl.12751
Hilty, J., Muller, B., Pantin, F. and Leuzinger, S., 2021. Plant growth: the What, the How, and the Why. New Phytologist, 232(1), pp.25–41. https://doi.org/10.1111/nph.17610
Hohnjec, N., Perlick, A.M., Pühler, A. and Küster, H., 2003. The Medicago truncatula sucrose synthase gene MtSucS1 is activated both in the infected region of root nodules and in the cortex of roots colonized by arbuscular mycorrhizal fungi . Molecular Plant-Microbe Interactions, 16(10), pp.903–915. https://doi.org/10.1094/mpmi.2003.16.10.903
Ivanov, S., Austin, J., Berg, R.H. and Harrison, M.J., 2019. Extensive membrane systems at the host–arbuscular mycorrhizal fungus interface. Nature Plants, 5(2), pp.194–203. https://doi.org/10.1038/s41477-019-0364-5
Janos, D.P., 2007. Plant responsiveness to mycorrhizas differs from dependence upon mycorrhizas. Mycorrhiza, 17(2), pp.75–91. https://doi.org/10.1007/s00572-006-0094-1
Jiang, Y., Wang, W., Xie, Q., Liu, N., Liu, L., Wang, D., Zhang, X., Yang, C., Chen, X., Tang, D. and Wang, E., 2017. Plants transfer lipids to sustain colonization by mutualistic mycorrhizal and parasitic fungi. Science, 356 (6343), pp.1172–1175. https://doi.org/10.1126/science.aam9970
Kameoka, H. and Gutjahr, C., 2022. Functions of lipids in development and reproduction of arbuscular mycorrhizal fungi. Plant and Cell Physiology, 63(10), pp.1356–1365. https://doi.org/10.1093/pcp/pcac113
Kameoka, H., Tsutsui, I., Saito, K., Kikuchi, Y., Handa, Y., Ezawa, T., Hayashi, H., Kawaguchi, M. and Akiyama, K., 2019. Stimulation of asymbiotic sporulation in arbuscular mycorrhizal fungi by fatty acids. Nature Microbiology, 4(10), pp.1654–1660. https://doi.org/10.1038/s41564-019-0485-7
Kaschuk, G., Kuyper, T.W., Leffelaar, P.A., Hungria, M. and Giller, K.E., 2009. Are the rates of photosynthesis stimulated by the carbon sink strength of rhizobial and arbuscular mycorrhizal symbioses? Soil Biology and Biochemistry, 41(6), pp.1233–1244. https://doi.org/10.1016/j.soilbio.2009.03.005
Khatami, M.H., Barber, W. and de Haan, H.W., 2021. Using geometric criteria to study helix-like structures produced in molecular dynamics simulations of single amylose chains in water. RSC Advances, 11(20), pp.11992–12002. https://doi.org/10.1039/d1ra00071c
Kiers, E.T., Duhamel, M., Beesetty, Y., Mensah, J.A., Franken, O., Verbruggen, E., Fellbaum, C.R., Kowalchuk, G.A., Hart, M.M., Bago, A., Palmer, T.M., West, S.A., Vandenkoornhuyse, P., Jansa, J. and Bücking, H., 2011. Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science, 333(6044), pp. 880–882. https://doi.org/10.1126/science.1208473
Kiers, T. and van der Heijden, M., 2006. Mutualistic stability in the arbuscular mycorrhizal symbosis. Ecology, 87(7), pp. https://doi.org/10.1890/0012-9658(2006)87[1627:MSITAM]2.0.CO;2
Koch, K., 2004. Sucrose metabolism: Regulatory mechanisms and pivotal roles in sugar sensing and plant development. Current Opinion in Plant Biology, 7(3), pp. 235–246. https://doi.org/10.1016/j.pbi.2004.03.014
Kodama, K., Rich, M.K., Yoda, A., Shimazaki, S., Xie, X., Akiyama, K., Mizuno, Y., Komatsu, A., Luo, Y., Suzuki, H., Kameoka, H., Libourel, C., Keller, J., Sakakibara, K., Nishiyama, T. and Nakagawa, T., 2022. An ancestral function of strigolactones as symbiotic rhizosphere signals. 13, 3974. https://doi.org/10.1038/s41467-022-31708-3
Kokkoris, V. and Hart, M., 2019. In vitro propagation of arbuscular mycorrhizal fungi may drive fungal evolution. Frontiers in Microbiology, 10:2420. https://doi.org/10.3389/fmicb.2019.02420
Konvalinková, T., Püschel, D., ?ezá?ová, V., Gryndlerová, H. and Jansa, J., 2017. Carbon flow from plant to arbuscular mycorrhizal fungi is reduced under phosphorus fertilization. Plant and Soil, 419, pp. 319–333, https://doi.org/10.1007/s11104-017-3350-6
Lemoine, R., Camera, S. La, Atanassova, R., Dédaldéchamp, F., Allario, T., Pourtau, N., Bonnemain, J.-L., Laloi, M., Coutos-Thévenot, P., Maurousset, L., Faucher, M., Girousse, C., Lemonnier, P., Parrilla, J. and Durand, M., 2013. Source-to-sink transport of sugar and regulation by environmental factors. Frontiers in Plant Science, 4:272. https://doi.org/10.3389/fpls.2013.00272
Liao, D., Sun, C., Liang, H., Wang, Y., Bian, X., Dong, C., Niu, X., Yang, M., Xu, G., Chen, A. and Wu, S., 2022. SlSPX1-SlPHR complexes mediate the suppression of arbuscular mycorrhizal symbiosis by phosphate repletion in tomato. The Plant Cell, 34: pp. 4045–4065. https://doi.org/10.1093/plcell/koac212
Lin, I.W., Sosso, D., Chen, L.Q., Gase, K., Kim, S.G., Kessler, D., Klinkenberg, P.M., Gorder, M.K., Hou, B.H., Qu, X.Q., Carter, C.J., Baldwin, I.T. and Frommer, W.B., 2014. Nectar secretion requires sucrose phosphate synthases and the sugar transporter SWEET9. Nature, 508(7497), pp.546–549. https://doi.org/10.1038/nature13082.
Liu, J.J., Chen, J., Xie, K., Tian, Y., Yan, A., Liu, J.J., Huang, Y., Wang, S., Zhu, Y., Chen, A. and Xu, G., 2020. A mycorrhiza-specific H+-ATPase is essential for arbuscule development and symbiotic phosphate and nitrogen uptake. Plant Cell and Environment, 43(4), pp. 1069–1083. https://doi.org/10.1111/pce.13714
Lü, L.-H., Zou, Y.-N. and Wu, Q.-S., 2018. Relationship between arbuscular mycorrhizas and plant growth: Improvement or depression? In: B. Giri, R. Prasad and A. Varma, eds. Root biology. Cham: Elsevier. pp.451–464. https://doi.org/10.1007/978-3-319-75910-4_18
Lu, W., Tang, X., Huo, Y., Xu, R., Qi, S., Huang, J., Zheng, C. and Wu, C., 2012. Identification and characterization of fructose 1,6-bisphosphate aldolase genes in Arabidopsis reveal a gene family with diverse responses to abiotic stresses. Gene, 503(1), pp.65–74. https://doi.org/10.1016/j.gene.2012.04.042
Luginbuehl, L.H., Menard, G.N., Kurup, S., Van Erp, H., Radhakrishnan, G. V., Breakspear, A., Oldroyd, G.E.D. and Eastmond, P.J., 2017. Fatty acids in arbuscular mycorrhizal fungi are synthesized by the host plant. Science, 356(6343), pp.1175–1178. https://doi.org/10.1126/science.aan0081
Lunn, J.E. and ap Rees, T., 1990. Apparent equilibrium constant and mass-action ratio for sucrose-phosphate synthase in seeds of Pisum sativum . Biochemical Journal, 267(3), pp.739–743. https://doi.org/10.1042/bj2670739
Ma, Y., Gao, G., Wang, S., Ren, H., Liu, Z., Chen, Y., Guo, Q. and Gu, J., 2024. Divergent arbuscular mycorrhizal growth responses in woody and herbaceous plants across inoculum richness. Environmental and Experimental Botany, 224, p.105811. https://doi.org/10.1016/j.envexpbot.2024.105811
Ma, Y., Yin, Z. and Ye, J., 2017. Lipid biosynthesis and regulation in Jatropha, an emerging model for woody energy plants. pp.113–127. https://doi.org/10.1007/978-3-319-49653-5_7
Maillet, F., Poinsot, V., André, O., Puech-Pagés, V., Haouy, A., Gueunier, M., Cromer, L., Giraudet, D., Formey, D., Niebel, A., Martinez, E.A., Driguez, H., Bécard, G. and Dénarié, J., 2011. Fungal lipochitooligosaccharide symbiotic signals in arbuscular mycorrhiza. Nature, 469(7328), pp.58–64. https://doi.org/10.1038/nature09622
Maloney, V.J., Park, J.Y., Unda, F. and Mansfield, S.D., 2015. Sucrose phosphate synthase and sucrose phosphate phosphatase interact in planta and promote plant growth and biomass accumulation. Journal of Experimental Botany, 66(14), pp.4383–4394. https://doi.org/10.1093/jxb/erv101
Manck-Götzenberger, J. and Requena, N., 2016. Arbuscular mycorrhiza symbiosis induces a major transcriptional reprogramming of the potato SWEET sugar transporter family. Frontiers in Plant Science, 7:487, pp.1–14. https://doi.org/10.3389/fpls.2016.00487
Micallef, B., Haskins, K., Vanderveer, P., Roh, K.-S., Shewmaker, C. and Sharkey, T., 1995. Altered photosynthesis, flowering, and fruiting in transgenic tomato plants that have an increased capacity for sucrose synthesis. Planta, 196(2), pp.327–334. https://doi.org/10.1007/BF00201392
Park, Y.J., Park, Y.J. and Park, C.M., 2023. Environmentally adaptive reshaping of plant photomorphogenesis by karrikin and strigolactone signaling. (Figure 1). https://doi.org/10.1111/jipb.13602
Pfeffer, P.E., Douds, D.D., Bécard, G. and Shachar-Hill, Y., 1999. Carbon uptake and the metabolism and transport of lipids in an arbuscular mycorrhiza. Plant Physiology, 120(2), pp.587–598. https://doi.org/10.1104/pp.120.2.587
Poorter, H. and Nagel, O., 2000. The role of biomass allocation in the growth response of plants to different levels of light, CO2, nutrients and water: a quantitative review. Australian Journal of Plant Physiology, 27(12), pp. 595–607. https://doi.org/10.1071/PP99173_CO
Preiss, J., 1984. Starch, sucrose biosynthesis and partition of carbon in plants are regulated by orthophosphate and triose-phosphates. Trends in Biochemical Sciences, 9(1), pp.24–27. https://doi.org/10.1016/0968-0004(84)90043-4
Prescott, C.E., Grayston, S.J., Helmisaari, H.S., Kaštovská, E., Körner, C., Lambers, H., Meier, I.C., Millard, P. and Ostonen, I., 2020. Surplus Carbon Drives Allocation and Plant–Soil Interactions. Trends in Ecology and Evolution, 35(12), pp.1110–1118. https://doi.org/10.1016/j.tree.2020.08.007
Proels, R.K. and Roitsch, T., 2009. Extracellular invertase LIN6 of tomato : a pivotal enzyme for integration of metabolic , hormonal , and stress signals is regulated by a diurnal rhythm. 60(6), pp.1555–1567. https://doi.org/10.1093/jxb/erp027
Rich, M.K., Nouri, E., Courty, P.E. and Reinhardt, D., 2017. Diet of Arbuscular Mycorrhizal Fungi: Bread and Butter? Trends in Plant Science, 22(8), pp.652–660. https://doi.org/10.1016/j.tplants.2017.05.008
Rich, M.K., Schorderet, M. and Reinhardt, D., 2014. The role of the cell wall compartment in mutualistic symbioses of plants. Frontiers in Plant Science, 5:238, pp.1–15. https://doi.org/10.3389/fpls.2014.00238
Rodrigues, J., Inzé, D., Nelissen, H. and Saibo, N.J.M., 2019. Source–Sink regulation in crops under water deficit. Trends in Plant Science, 24(7), pp.652–663. https://doi.org/10.1016/j.tplants.2019.04.005
Roth, R. and Paszkowski, U., 2017. Plant carbon nourishment of arbuscular mycorrhizal fungi. Current Opinion in Plant Biology, 39, pp.50–56. https://doi.org/10.1016/j.pbi.2017.05.008
Rufty, T.W. and Huber, S.C., 1983. Changes in starch formation and activities of sucrose phosphate synthase and cytoplasmic fructose-1,6-bisphosphatase in response to source-sink alterations. Plant Physiology, 72(2), pp.474–480. https://doi.org/10.1104/pp.72.2.474
Rui, W., Mao, Z. and Li, Z., 2022. The roles of phosphorus and nitrogen nutrient transporters in the arbuscular mycorrhizal symbiosis. International Journal of Molecular Sciences, 23(19). https://doi.org/10.3390/ijms231911027
Ruytinx, J., Kafle, A., Usman, M., Coninx, L., Zimmermann, S.D. and Garcia, K., 2020. Micronutrient transport in mycorrhizal symbiosis; zinc steals the show. Fungal Biology Reviews, 34(1), pp.1–9. https://doi.org/10.1016/j.fbr.2019.09.001
Salmeron?Santiago, I.A., Martínez?Trujillo, M., Valdez?Alarcón, J.J., Pedraza?Santos, M.E., Santoyo, G., López, P.A., Larsen, J., Pozo, M.J. and Chávez?Bárcenas, A.T., 2023. Carbohydrate and lipid balances in the positive plant phenotypic response to arbuscular mycorrhiza: increase in sink strength. Physiologia Plantarum, 175(1). https://doi.org/10.1111/ppl.13857
Salmeron?Santiago, I.A., Martínez?Trujillo, M., Valdez?Alarcón, J.J., Pedraza?Santos, M.E., Santoyo, G., Pozo, M.J. and Chávez?Bárcenas, A.T., 2022. An updated review on the modulation of carbon partitioning and allocation in arbuscular mycorrhizal plants. Microorganisms, 10(1), pp.1–20. https://doi.org/10.3390/microorganisms10010075
Sawers, R.J.H., Svane, S.F., Quan, C., Grønlund, M., Wozniak, B., Gebreselassie, M.N., González-Muñoz, E., Chávez Montes, R.A., Baxter, I., Goudet, J., Jakobsen, I. and Paszkowski, U., 2017. Phosphorus acquisition efficiency in arbuscular mycorrhizal maize is correlated with the abundance of root-external hyphae and the accumulation of transcripts encoding PHT1 phosphate transporters. New Phytologist, 214(2), pp.632–643. https://doi.org/10.1111/nph.14403
Schaarschmidt, S., Gonzalez, M.-C., Roitsch, T., Strack, D., Sonnewald, U. and Hause, B., 2007a. Regulation of Arbuscular Mycorrhization by Carbon. The Symbiotic Interaction Cannot Be Improved by Increased Carbon Availability Accomplished by Root-Specifically Enhanced Invertase Activity. PLANT PHYSIOLOGY, 143(4), pp.1827–1840. https://doi.org/10.1104/pp.106.096446
Schaarschmidt, S., Kopka, J., Ludwig-mu, J., Hause, B., Mu, A. and Universita, T., 2007b. Regulation of arbuscular mycorrhization by apoplastic invertases : enhanced invertase activity in the leaf apoplast affects the symbiotic interaction. 51, pp. 390–405. https://doi.org/10.1111/j.1365-313X.2007.03150.x
Schaarschmidt, S., Roitsch, T. and Hause, B., 2006. Arbuscular mycorrhiza induces gene expression of the apoplastic invertase LIN6 in tomato (Lycopersicon esculentum) roots. 57(15), pp.4015–4023. https://doi.org/10.1093/jxb/erl172
Schubert, A., Allara, P. and Morte, A., 2003. Cleavage of sucrose in roots of soybean (Glycine max) colonized by an arbuscular mycorrhizal fungus. New Phytologist, 161(2), pp.495–501. https://doi.org/10.1046/j.1469-8137.2003.00965.x
Siddappaji, M.H., Scholes, D.R., Krishnankutty, S.M., Calla, B., Clough, S.J., Zielinski, R.E. and Paige, K.N., 2015. The role of invertases in plant compensatory responses to simulated herbivory. BMC Plant Biology, 15(1), pp.1–12. https://doi.org/10.1186/s12870-015-0655-6
Smith, S. and Read, D., 2008. Mycorrhizal Symbiosis. https://doi.org/10.1016/B978-0-12-370526-6.X5001-6
Smith, S.E., Jakobsen, I., Grønlund, M. and Smith, F.A., 2011. Roles of arbuscular mycorrhizas in plant phosphorus nutrition: Interactions between pathways of phosphorus uptake in arbuscular mycorrhizal roots have important implications for understanding and manipulating plant phosphorus acquisition. Plant Physiology, 156(3), pp.1050–1057. https://doi.org/10.1104/pp.111.174581
Stein, O. and Granot, D., 2019. An overview of sucrose synthases in plants. Frontiers in Plant Science, 10:95., pp.1–14. https://doi.org/10.3389/fpls.2019.00095
Sugiura, Y., Akiyama, R., Tanaka, S., Yano, K., Kameoka, H., Marui, S., Saito, M., Kawaguchi, M., Akiyama, K. and Saito, K., 2020. Myristate can be used as a carbon and energy source for the asymbiotic growth of arbuscular mycorrhizal fungi. Proceedings of the National Academy of Sciences of the United States of America, 117(41), pp.25779–25788. https://doi.org/10.1073/pnas.2006948117
Tamayo, E., Figueira-Galán, D., Manck-Götzenberger, J. and Requena, N., 2022. Overexpression of the potato monosaccharide transporter StSWEET7a promotes root colonization by symbiotic and pathogenic fungi by increasing root sink strength. Frontiers in Plant Science, 13:837231, pp.1–18. https://doi.org/10.3389/fpls.2022.837231
Tanaka, S., Hashimoto, K., Kobayashi, Y., Yano, K., Maeda, T., Kameoka, H., Ezawa, T., Saito, K., Akiyama, K. and Kawaguchi, M., 2020. Asymbiotic mass production of the arbuscular mycorrhizal fungus Rhizophagus clarus. bioRxiv, https://doi.org/10.1101/2020.12.25.424379
Tetlow, I.J. and Bertoft, E., 2020. A review of starch biosynthesis in relation to the building block-backbone model. International Journal of Molecular Sciences, 21(19), pp.1–37. https://doi.org/10.3390/ijms21197011
Tran, B.T.T., Watts-Williams, S.J. and Cavagnaro, T.R., 2019. Impact of an arbuscular mycorrhizal fungus on the growth and nutrition of fifteen crop and pasture plant species. Functional Plant Biology, 46(8), pp.732–742. https://doi.org/10.1071/FP18327
Tsai, C.H., Miller, A., Spalding, M. and Rodermel, S., 1997. Source strength regulates an early phase transition of tobacco shoot morphogenesis. Plant Physiology, 115(3), pp.907–914. https://doi.org/10.1104/pp.115.3.907
van’t Padje, A., Werner, G.D.A. and Kiers, E.T., 2021. Mycorrhizal fungi control phosphorus value in trade symbiosis with host roots when exposed to abrupt ‘crashes’ and ‘booms’ of resource availability. New Phytologist, 229(5), pp.2933–2944. https://doi.org/10.1111/nph.17055
Walder, F. and Van Der Heijden, M.G.A., 2015. Regulation of resource exchange in the arbuscular mycorrhizal symbiosis. Nature Plants, 1, 15159 pp.1–7. https://doi.org/10.1038/nplants.2015.159
Wewer, V., Brands, M. and Dörmann, P., 2014. Fatty acid synthesis and lipid metabolism in the obligate biotrophic fungus Rhizophagus irregularis during mycorrhization of Lotus japonicus. Plant Journal, 79(3), pp.398–412. https://doi.org/10.1111/tpj.12566
Wipf, D., Krajinski, F., van Tuinen, D., Recorbet, G. and Courty, P.E., 2019. Trading on the arbuscular mycorrhiza market: from arbuscules to common mycorrhizal networks. New Phytologist, 223(3), pp.1127–1142. https://doi.org/10.1111/nph.15775
Wright, D.P., Read, D.J. and Scholes, J.D., 1998. Mycorrhizal sink strength influences whole plant carbon balance of Trifolium repens L. Plant, Cell and Environment, 21(9), pp.881–891. https://doi.org/10.1046/j.1365-3040.1998.00351.x
Xu, X., Yang, Y., Liu, C., Sun, Y., Zhang, T., Hou, M., Huang, S. and Yuan, H., 2019. The evolutionary history of the sucrose synthase gene family in higher plants. BMC Plant Biology, 19(1), pp.1–14. https://doi.org/10.1186/s12870-019-2181-4
Zhang, Q., Blaylock, L.A. and Harrison, M.J., 2010. Two Medicago truncatula half-ABC transporters are essential for arbuscule development in arbuscular mycorrhizal symbiosis. Plant Cell, 22(5), pp.1483–1497. https://doi.org/10.1105/tpc.110.074955
URN: http://www.revista.ccba.uady.mx/urn:ISSN:1870-0462-tsaes.v29i1.65742
DOI: http://dx.doi.org/10.56369/tsaes.6574
Copyright (c) 2026 Ana Tztzqui Chávez Bárcenas

This work is licensed under a Creative Commons Attribution 4.0 International License.
